Revolutionizing Healthcare with Convenient and Accurate At-Home Tests
About Our Labs and Science: Home Diagnostic Testing
At Healthyr, we’re dedicated to improving healthcare with developing cutting-edge technology that allows patients to perform diagnostic tests in the comfort of their own homes. People can now monitor their health more easily and conveniently than ever before.
Our research is driven by the belief that healthcare should be accessible and personalized for every individual. That’s why we’re committed to developing safe, easy-to-use testing solutions that can be taken by anyone, regardless of their location or circumstance.

The Science Behind the Test
What is Dried Blood Spot Testing?
Dried Blood Spot (DBS) Testing is a simple and effective way to check important health markers without the need for a blood draw or lab visit. This established technology has been in use since the 1960’s in areas such as neonatal care and for health testing in very remote locations. Healthyr has expanded this technology to test a wide range of health biomarkers, allowing you to check various aspects of your health from the comfort of your own home.
Our tests utilize an ADX card which contains a special type of paper that filters the blood solids from the blood plasma.
You collect your sample by pricking your finger with the provided lancet, and place a few drops of blood onto the card.
Once your sample is dry, you can mail it to the lab, where it will be reconstituted and tested on reliable central laboratory equipment. From there we can test for things like proteins, vitamins, minerals, hormones, and key indicators of organ and body system health.
Is DBS Testing reliable?
Multiple studies support the use of DBS and other self-collection methods for various biomarkers. The accuracy of DBS results is designed to work in a similar way to clinically-collected samples. The tests offered by Healthyr and those ordered in a physician’s office are comparable, and our partner labs’ testing methods and instruments go through a thorough validation process to ensure accuracy, precision, and clinical correlation. Before approving any of Healthyr’s tests, studies are conducted to ensure that the results from our DBS samples compare to traditional blood draw results within nationally-accepted limits of variation.
About Our Labs
Our mission is to empower individuals to take control of their health by offering convenient, reliable, and accurate testing options. We do that by working with the most highly qualified laboratories in the country. Our team of experienced laboratory professionals follows strict safety protocols to ensure the accuracy and reliability of your test results.
We hold ourselves accountable for:
Personal Protective Equipment (PPE)
Our laboratory personnel wear appropriate PPE such as gloves, lab coats, and eye protection to protect themselves from potential hazards like chemicals, biological agents, and radiation.
Chemical Safety
Our labs follow strict chemical safety protocols to ensure the safe handling, storage, and disposal of hazardous chemicals. This includes proper labeling, the use of appropriate containment and ventilation systems, and regular safety audits and inspections.
Biological Safety
Our labs follow strict biological safety protocols to ensure the safe handling and disposal of biological agents such as blood, tissue, and other specimens. This includes properly using biohazard containment systems, disposal of infectious waste, and regular safety training and inspections.
Equipment Safety
Our labs ensure that all laboratory equipment is properly maintained, calibrated, and validated to ensure accurate and reliable test results. This includes regular equipment inspections, and maintenance, as well as regular safety training for laboratory personnel.
Quality Control
Our labs follow rigorous quality control protocols to ensure the accuracy and reliability of test results. This includes regular internal and external quality control checks, participation in proficiency testing programs, and adherence to recognized standards such as CLIA and GLP.
Certifications, Safety Protocols, and Accreditations
CAP-Accreditation
The College of American Pathologists (CAP) is considered the gold standard in lab testing quality regulation in the United States. They use strict guidelines and standards to ensure that CAP-accredited labs maintain the accuracy of test results, ensure accurate patient diagnosis, meet or exceed standards for patient care and safety, stay up-to-date with changes in laboratory medicine and technological best practices, and work with laboratory staff to offer ongoing development and training. Their annual checklists for accreditation are compiled with input from over 500 certified pathologists to achieve the highest quality lab operations and laboratory inspections occur bi-annually to ensure compliance.
CLIA-Certification
CLIA stands for Clinical Laboratory Improvement Amendments. Their certification program regulates clinical laboratories in the United States to ensure that they are producing accurate and reliable test results.
The CLIA-Certification program sets national standards for laboratory testing quality and safety. Laboratories that perform testing on human specimens, such as blood, urine, or tissue, must meet certain requirements and pass inspections to receive and maintain their certification.
The CLIA program applies to all types of laboratory testing, including tests performed in hospitals, clinics, physician offices, and research facilities. The program also requires laboratories to have trained personnel, quality control procedures, and appropriate record-keeping and reporting practices.
Good Laboratory Practices
Good Laboratory Practices (GLP) is a set of guidelines that ensure the quality, reliability, and integrity of non-clinical laboratory studies in accordance with regulatory submission guidelines. GLP covers all aspects of laboratory testing, from the study design to the final report, to ensure that the data generated is of high quality and scientifically sound.
The main purpose of GLP is to ensure that laboratory studies are conducted in a consistent, reliable, and traceable manner. This includes things like maintaining accurate records, ensuring that instruments and equipment are properly calibrated and maintained, and providing appropriate training to laboratory personnel.
GLP applies to all non-clinical laboratory studies that are submitted to regulatory agencies, such as the US Food and Drug Administration (FDA), the Environmental Protection Agency (EPA), and the European Medicines Agency (EMA).
By following GLP, laboratories can ensure that the data generated from their studies are reliable and can be used to support regulatory submissions. This helps to ensure the safety and effectiveness of products that are marketed to the public, such as drugs, medical devices, and pesticides.
High-Complexity Laboratory
A high-complexity laboratory has been evaluated and certified by an accrediting organization to meet certain quality standards for performing high-complexity testing. The term “high complexity” refers to the types of tests that are performed in the laboratory. These tests typically require a high degree of scientific and technical knowledge, as well as specialized equipment and procedures. High-complexity tests are used to diagnose and monitor complex diseases and conditions and guide treatment decisions.
Accreditation is a voluntary process in which a laboratory undergoes a comprehensive evaluation of its facilities, equipment, personnel, procedures, and quality control systems. Accrediting organizations, such as the College of American Pathologists (CAP) or the Clinical Laboratory Improvement Amendments (CLIA) program, review the laboratory’s performance against established standards and guidelines to determine if it meets the requirements for accreditation.
An accredited high-complexity laboratory demonstrates its commitment to quality and accuracy in performing high-complexity testing. This provides assurance to patients, healthcare providers, and regulatory agencies that the laboratory is capable of producing reliable and accurate test results. In addition, accreditation can improve the reputation and credibility of the laboratory and may be required for participation in certain healthcare programs or insurance networks.
Physician-Supported At-Home Testing for Better Health Management
Empowering You with Personalized Results Reviewed by Board-Certified Physicians


Physician Involvement
At Healthyr, we understand the importance of having real doctors involved in the testing process. That’s why we work with a network of independent board-certified physicians who review your lab requisition and approve eligible orders. They also review your final test results and offer consultation services for results that require significant action, such as a positive STI result. With Healthyr, you can feel confident that you’re getting reliable and accurate results that are interpreted by experienced medical professionals.
Easy-to-Understand Results
We believe that everyone deserves personalized information and education that empowers them to take control of their health. That’s why we offer a downloadable version of your results, in a format that your physician is used to seeing when reviewing lab results. This makes it easier for you to share your results with your doctor and ask any questions you have about your medical care.




Labwork Prescription
In many states, consumers are required to have a prescription from a board-certified medical doctor to order their own tests. At Healthyr, we work with a third-party independent network of physicians who provide authorization, review, and critical value consultation services for our customers related to their tests. Once you request a test, the independent physician reviews your request and determines whether the test is appropriate for you. If it’s not, you’ll receive a refund. If it is, the physician orders the test, and the lab sends the results to the physician for review. After the physician approves the results, they’re released and delivered to you through our secure HIPAA-protected online results dashboard.
Critical Value and Nationally Notifiable Diseases
At Healthyr, we take all results seriously, especially those with predetermined “critical values.” Our physician network maintains a list of these values and triages all out-of-range results. If you have a critical value result or a positive result for an STI, a physician from your state will contact you directly. With Healthyr’s physician-supported at-home testing, you can take control of your health and get the personalized information you need to make informed decisions about your well-being.


Keeping Your Information Secure
At Healthyr, we take your privacy and security very seriously. We use unique barcode IDs for every sample received by our partner labs. This ID ensures that your results are accurately delivered to you and reduces the chance of any errors caused by human mistakes.
Healthyr’s labs follow strict protocols to process your samples, guaranteeing the highest level of accuracy and quality.
We use validated extraction protocols to ensure the end-to-end process produces the most accurate results possible.
HIPAA Compliance
HIPAA Compliance is a set of rules and regulations that healthcare organizations must follow to protect your personal health information. HIPAA stands for Health Insurance Portability and Accountability Act, a law passed in the United States to ensure patient privacy.
Healthyr has policies and procedures in place to protect your privacy and trains its employees to follow them.
HITECH Security Standards
The HITECH Security Standards are regulations designed to protect your sensitive health information from theft or unauthorized disclosure. The standards require healthcare organizations and their business associates to implement specific security measures, such as encrypting data and restricting access to authorized personnel.
By following these standards, Healthyr can help ensure that your personal and medical information remains confidential and secure.
We’re proud to be at the forefront of the at-home diagnostic testing field. Our goal is to continually make a real difference in people’s lives. By providing patients with the tools they need to monitor their health more closely, we empower them to take control of their well-being and make informed decisions about their care.
Thank you for choosing Healthyr as your partner in healthcare. We’re dedicated to continuing to innovate diagnostic testing and health solutions that make healthcare more convenient, accurate, and accessible for everyone.
Introducing Auria At-Home Breast Screening
The Auria at-home breast cancer screening test empowers you to take control of your health. By providing a simple and convenient test that can be done from the comfort of your own home, Auria offers a truly innovative solution to detecting potential signs of breast cancer, with the power of tears.


Why tears?
Protein biomarkers found in tears
Auria analyzes the protein biomarkers found in your tears. Unlike DNA, proteins undergo changes in response to different molecular events that occur in the body. These proteins are significant in the initial stages of inflammation and may increase due to irregularities in your breast tissue.
Breast abnormalities could be caused by various factors and can take different forms, like fibrocystic changes, early tumor formation, cysts, and calcifications. The Auria test can indicate changes in protein biomarkers, which means further assessment of your breast tissue may be necessary. While the test does not confirm the presence of breast cancer, like a mammogram, it can provide valuable information for further evaluation.



Backed by research
Tears, the transparent liquid produced by our eyes, have the potential to offer significant insights into our health. This often-overlooked fluid could prove invaluable in the early detection of diseases like breast cancer.
Tear composition includes proteins, lipids, small molecules, and even genetic material, that might serve as indicators of underlying health conditions. The process of collecting tear samples is simple, painless, and doesn’t require invasive procedures.
This approach stands in contrast to blood collection, which can be more invasive. Researchers are actively comparing the content of tears to that of blood to establish tears as a promising source of biomarkers, potentially revolutionizing disease detection and diagnosis.
What We Found
Tears could be the key to detecting diseases quickly and conveniently. Scientists are exploring the potential of using tears for biomarker research, which could revolutionize the way we detect diseases. The next step is to study patients with specific diseases to understand the potential of tear biomarkers for diagnosis. Exciting times lie ahead, and who knows, maybe one day we’ll be able to detect diseases just by shedding a tear!
Trust and Accuracy
The Auria test has been developed using a wealth of scientific research. It’s important to note that while this test shows promise, it’s not a definitive diagnosis. However, it can serve as a valuable first step in identifying potential risks. The sensitivity and specificity of the test – its ability to correctly identify those with and without breast cancer – have been carefully evaluated and refined through extensive studies.
Empowering Early Detection
The power of the Auria test lies in its ability to provide early indications of potential breast health concerns. By using tears as a non-invasive and accessible source of information, this test aims to empower individuals to take charge of their health and engage in proactive conversations with healthcare professionals.


A step forward in health care
The Auria at-home breast screening test is a significant milestone in the healthcare industry. It is a revolutionary innovation that utilizes tears to extract valuable information for the early detection of breast health issues. While no screening test is perfect, Auria represents a significant step toward making breast health monitoring proactive, easy, and accessible. It empowers individuals to take charge of their health by providing them with vital information to prioritize their well-being with confidence.
Next steps
If the Auria test suggests a potential concern, following up with your healthcare provider is essential. They can recommend further examinations or screenings to confirm the results. Remember, the Auria test complements existing screening methods and enhances your breast health journey.


Frequently Asked Questions
Auria’s proprietary algorithm uses the concentrations of S100A8 and S100A9 determined from the tear sample, together with age, to produce the score.
Namida Lab (developer of Auria®) has 3 ongoing clinical trials. The following are Namida’s current clinical trial numbers: NCT05268224-DB; NCT05223218; NCT04928014
Genetic screening is used to identify mutations in the BRCA1 and BRCA2 genes and can be a great tool, particularly for women with a family history of breast cancer. However, genetic screening is expensive and these genes only account for 15-20% of all breast cancers. 89% of breast cancer occurs in women with no family history.
Auria is a lab-developed test (LDT) that has been validated in Namida Lab’s high-complexity Clinical Laboratory Improvement Amendments (CLIA) lab under the supervision of our Medical Director. An LDT is a test that is designed, developed, and performed within a single laboratory, regulated by the Centers for Medicare & Medicaid Services (CMS), through the Clinical Laboratory Improvement Amendments (CLIA). Currently, the FDA exercises enforcement discretion over LDTs. Certification of the laboratory is required under CLIA to ensure the validity and quality of the test.
Unfortunately, we have not been able to conduct a study looking at our markers for reoccurrence in breast cancer so without that data is difficult to say for sure. While we did have triple negative cases in our breast cancer clinical data set we used for validating the test in our CLIA lab that were all accurately classified, the overall number was quite small and they were newly diagnosed cases not under treatment. It’s very likely long-term treatment for metastatic breast cancer could impact the expression of our markers.
Yes, your kit does expire. Your kit will have a minimum expiration date of 6 months. The expiration date can be found on the label on the bottom of your Auria box. Any samples that are delivered to the lab after the expiration date will not be processed, and we are unable to provide a replacement kit.
The breast health consultation provided with every test kit regardless of risk score, is with a breast health expert that is either a Nurse Practitioner or Registered Nurse.
Your Auria consultation is an engaging, informative session with one of our Breast Health Specialists. They provide insights based on your unique health history, answer questions, and guide you toward optimal breast health outcomes. You will gain early access to expert insights that are usually offered post-diagnosis. Your consultation is personalized and focused on your unique needs. All consultations are included with the Auria test kit.
Once your results are ready, you’ll be able to schedule a consultation with our Breast Health Specialists (BHS) through an email link or through your dashboard. At your scheduled appointment time, you will meet with a BHS via a Zoom link that is emailed to you. During the consultation, the BHS will review your results and the information you provided in your breast health survey and will guide you through potential next steps. You will understand the impact of modifiable risk factors on your breast health, and you will have an open session time for you to ask any lingering questions you may have.
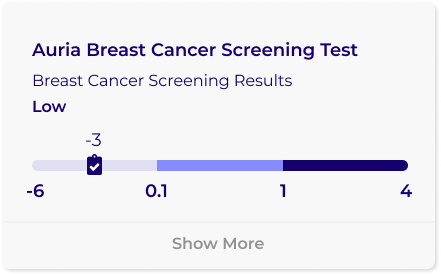
Your test results will provide you with a score and classification of low, medium, or high. A medium or high classification does not mean you have breast cancer, but rather the score produced from your sample could be elevated due to a breast abnormality.
Auria determines the concentration of two proteins from your tear sample. These two proteins are part of the early inflammatory process and could be elevated due to malignancy but can also be elevated due to benign masses. These concentrations are used, together with age, in our proprietary algorithm to calculate your AURIA SCORE.
The AURIA SCALE was developed using the scores from over 1000 clinical samples. The classifications of low, medium, and high were developed based on retrospective clinical analysis of follow-up imaging. Early data suggests scores in the medium or high category are associated with increased molecular events in the host tissue and should be evaluated by follow-up imaging in a timely manner.


